|
Next on the table is helium, whose atoms have two protons in the nucleus. For example, carbon has six protons and is atomic number 6. Every hydrogen atom has one proton in its nucleus. The number of protons in a nucleus determines the elements atomic number on the Periodic Table. When an organism dies, it stops taking in carbon-14, so the ratio of carbon-14 to carbon-12 in its remains, such as fossilized bones, will decline as carbon-14 decays gradually to nitrogen-14 2 ^2 2 squared. Hydrogen, at the upper left of the table, has an atomic number of 1. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The nucleus is composed of protons and neutrons. 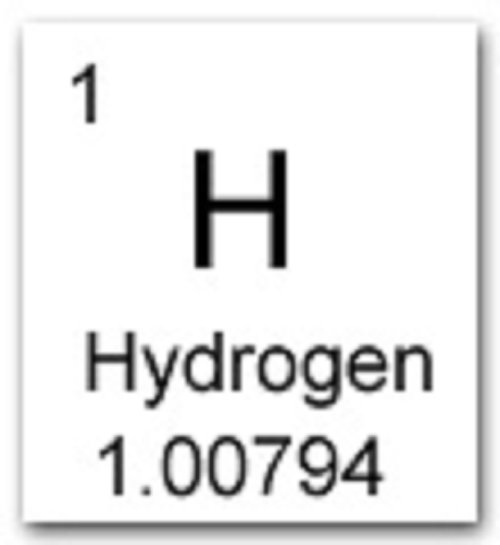
The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Hydrogen is a chemical element of the periodic table with chemical symbol H and atomic number 1 with an atomic weight of 1.0079 u and is classed as nonmetal and is part of group 1 (alkali metal). As animals eat the plants, or eat other animals that ate plants, the concentrations of carbon-14 in their bodies will also match the atmospheric concentration. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. As plants pull carbon dioxide from the air to make sugars, the relative amount of carbon-14 in their tissues will be equal to the concentration of carbon-14 in the atmosphere. For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. These forms of carbon are found in the atmosphere in relatively constant proportions, with carbon-12 as the major form at about 99%, carbon-13 as a minor form at about 1%, and carbon-14 present only in tiny amounts 1 ^1 1 start superscript, 1, end superscript. For example, carbon is normally present in the atmosphere in the form of gases like carbon dioxide, and it exists in three isotopic forms: carbon-12 and carbon-13, which are stable, and carbon-14, which is radioactive.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
